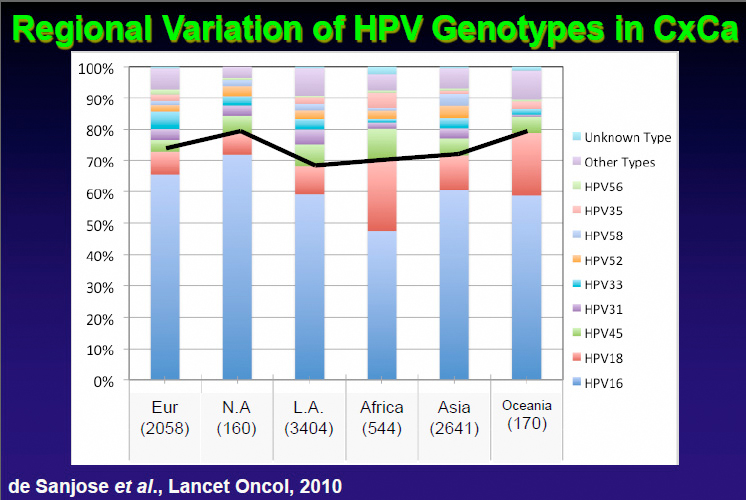
CÉDULA EN LA DIRECCIÓN GENERAL DE PROFESIONES (médico general): 1146192 | AUTORIZACIÓN PARA EJERCER LA ESPECIALIDAD DE GINECO-OBSTETRA DE LA D.G.P. No. AEIE-006714 (cédula federal de especialista) | CÉDULA ESTATAL DE MÉDICO GENERAL: 10736 (12-1) | CEDULA ESTATAL DE GINECÓLOGO (JALISCO): 129 (12-119) E.
Actividades Profesionales, Técnicas, Auxiliares y Especialidades. COFEPRIS-02-002-A. Establecimiento: Consultorio Médico Especialista en Ginecología. Ubicación: Guadalajara, Jalisco. Finalizado: 2023-02-08 Tipo de trámite: Alta. Número de ingreso: 2314102002A00015. Acuse de recibo.